Founding, Scaling, and Exiting a GMP-Compliant Contract Testing Laboratory

Results Snapshot
Our founder identified a market gap in quality control testing services and built a fully compliant contract testing laboratory from the ground up, achieving successful exit in 2024.
Key Outcomes
- Identified market opportunity in contract testing for pharmaceuticals, natural health products, and consumer goods through international industry experience.
- Raised capital through strategic equity partnerships and loans, including a landlord-investor arrangement that enabled facility customization.
- Designed and constructed a purpose-built GMP and ISO 17025-compliant laboratory facility from an empty commercial unit.
- Achieved regulatory excellence by passing Health Canada GMP (Drug Establishment License), ISO 17025, Biosafety Level II, Controlled Substance, and EuGMP audits.
- Scaled operations to serve global pharmaceutical clients, natural health product manufacturers, US dietary supplement companies, and consumer goods producers.
- Successfully exited the business through private sale in 2024, transitioning to found Terraforme Biosciences.
Project Context
After years of working in the corporate life sciences industry across the United States and Europe, our founder recognized a critical gap in the market: insufficient quality control testing capacity for pharmaceuticals, natural health products, dietary supplements, and certain consumer goods categories.
In 2017, he incorporated a new venture to address this unmet need. The vision was ambitious: build a contract testing laboratory capable of meeting the highest regulatory standards while remaining commercially competitive in a specialized market.
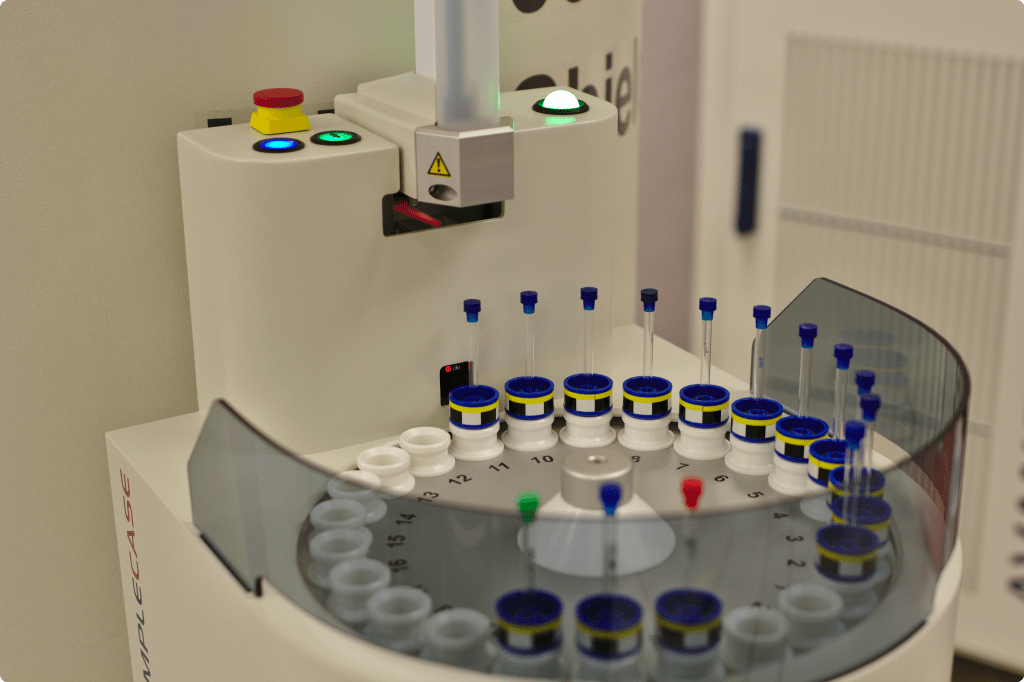
The challenge
Building a contract testing laboratory from inception to operational excellence presented multifaceted challenges:
Regulatory Complexity
Achieving simultaneous compliance with multiple regulatory frameworks—GMP (Drug Establishment License), ISO 17025 accreditation, Biosafety Level II certification, and Controlled Substance licensing—required designing quality and safety into every aspect of the facility and operations from day one.
Capital Efficiency
Balancing the need for state-of-the-art analytical equipment with the financial realities of a startup required strategic decisions about new versus refurbished equipment, vendor partnerships, and in-house validation capabilities.
Simultaneous Execution
The organization needed to execute multiple parallel workstreams: facility construction, Quality Management System (QMS) development, equipment procurement and qualification, method validation, team recruitment, and commercial pipeline development—all while maintaining readiness for regulatory audits.
Commercial Timing
Maintaining warm commercial leads during an extended setup phase required careful stakeholder management to ensure revenue generation could begin immediately upon facility completion.
What We Did
The founder orchestrated a comprehensive, methodical approach to laboratory establishment
Strategic Planning & Capitalization
The founder developed a detailed business plan and raised capital through a mixed-capital structure combining equity investment and loans. A strategic arrangement was negotiated whereby an investor purchased the commercial real estate and served as landlord, providing flexibility for extensive leasehold improvements critical to laboratory operations.
Team Assembly
Before breaking ground, the founder recruited a core leadership team spanning critical functional areas: analytical chemistry department lead, microbiology department lead, quality assurance lead, and operations lead. This ensured domain expertise was embedded in every design decision.
Facility Design & Construction
Starting with an empty commercial unit, the team conducted comprehensive analysis of equipment requirements, analytical processes, hazard assessments, and infrastructure needs. This analysis informed a design brief that defined all site requirements.
The design brief formed the basis of an RFQ process to select an architectural partner. The resulting architectural plans served dual purposes: permit applications and contractor RFQ documentation. Construction proceeded with full regulatory compliance as the foundational design principle.
Quality Management System Development
Concurrent with construction, the team developed a comprehensive QMS designed to meet GMP and ISO 17025 requirements. Recognizing that GMP compliance must be “designed in before shovels go in the ground,” the QMS development addressed:
- Change control and document control systems
- Standard Operating Procedures (SOPs), forms, and records management
- Human resources and training programs
- Facility maintenance plans
- Equipment IQ/OQ/PQ (Installation/Operational/Performance Qualification) protocols
- Computerized system validation per GAMP 5 guidelines for IT infrastructure
- Calibration procedures for all analytical equipment
- Biosafety Level II operational requirements
- Controlled substance handling protocols
Equipment Procurement & Qualification
As construction neared completion, the organization issued purchase orders for laboratory equipment across both departments
Microbiology: XL autoclave, multiple incubators, biosafety cabinets, and supporting equipment.
Analytical Chemistry: Induction Coupled Plasma Mass Spectrometer (ICP-MS) with microwave digestion, Gas Chromatography Flame Ionization Detector (GC-FID), High Pressure Liquid Chromatography with Diode Array Detector (HPLC-DAD), Liquid Chromatography Triple Quadrupole Mass Spectrometer (LC-MS/MS), and extensive standard laboratory instrumentation.
The team implemented a cost-optimization strategy, purchasing select equipment new from vendor partners while acquiring refurbished equipment for other applications. For refurbished equipment, the organization designed and executed IQ/OQ/PQ protocols in-house per GMP and GAMP 5 guidelines, ensuring validation rigor while controlling costs.
Method Development & Validation
Following equipment qualification, the laboratory executed method development and validation studies. The team planned studies accounting for the diverse sample matrices the laboratory would encounter: pharmaceutical formulations, natural health products, dietary supplements, personal care products, and environmental samples.
Method validation studies were conducted systematically, with results documented in formal validation reports. Final SOPs were then generated for operational use, creating a validated analytical toolkit ready for client samples.
Commercial Pipeline Management
Throughout the facility development phase, the founder maintained active engagement with prospective clients, managing expectations about timeline while preserving commercial relationships. This ensured a robust pipeline of business ready to activate upon facility completion.
Regulatory Audits & Expansion
The organization successfully passed all planned regulatory audits: GMP (Drug Establishment License), ISO 17025 accreditation, Biosafety Level II certification, and Controlled Substance licensing.
As the client base expanded to include European pharmaceutical companies, the laboratory underwent and successfully passed EuGMP audit by German regulatory inspectors, demonstrating compliance with international standards.
In response to growing demand for stability testing services, the laboratory later expanded capabilities to include real-time and accelerated stability studies with dedicated stability chambers.
Scaling & Diversification
The business scaled to serve a diverse client base across multiple sectors
- Global pharmaceutical manufacturers
- Natural health product companies
- US dietary supplement producers
- Personal care product manufacturers
- Consumer packaged goods companies
- Chemical manufacturers
- Facility water monitoring clients
- Environmental monitoring services
The Approach
The founder’s methodology combined regulatory foresight with operational pragmatism
Compliance-by-Design
Rather than retrofitting compliance, the organization embedded GMP, ISO 17025, biosafety, and controlled substance requirements into facility design, QMS development, and operational planning from inception.
Strategic Capital Deployment
The mixed approach to equipment procurement—combining new vendor-supported equipment with in-house validated refurbished equipment—optimized capital efficiency without compromising validation integrity.
Parallel Execution
By running facility construction, QMS development, team building, and commercial development as parallel workstreams rather than sequential phases, the organization compressed time-to-market and maintained stakeholder engagement.
Quality as Competitive Advantage
The decision to pursue the highest regulatory standards (including international EuGMP compliance) positioned the laboratory to serve premium clients and command sustainable margins in a competitive market.
Deliverables
1) Operational GMP & ISO 17025 Laboratory
A fully functional, multi-accredited contract testing facility serving pharmaceutical, natural health product, dietary supplement, and consumer goods sectors.
2) Comprehensive Quality Management System
A complete QMS meeting GMP, ISO 17025, Biosafety Level II, and Controlled Substance regulatory requirements, with validated computerized systems per GAMP 5.
3) Validated Analytical Capabilities
Qualified equipment and validated methods across microbiology and analytical chemistry departments, capable of handling diverse sample matrices.
4) Regulatory Track Record
Successful audit history including GMP (Drug Establishment License), ISO 17025, Biosafety Level II, Controlled Substance, and EuGMP inspections.
5) Diversified Client Portfolio
Revenue-generating relationships spanning pharmaceutical, natural health product, dietary supplement, personal care, consumer goods, chemical, and environmental monitoring sectors.
6) Successful Exit
Private sale transaction in 2024 enabling the founder’s transition to Terraforme Biosciences.
Lessons Learned
Design for Compliance, Not Retrofit: Building GMP and ISO 17025 compliance into facility design and QMS development from day one prevents costly retrofits and accelerates regulatory approval timelines.
Team Before Infrastructure: Recruiting technical leadership early ensures domain expertise informs every design decision, from facility layout to equipment selection to method development priorities.
Capital Efficiency Through Validation Expertise: In-house capability to design and execute IQ/OQ/PQ protocols for refurbished equipment provides significant cost savings while maintaining regulatory compliance.
Commercial Patience Pays Off: Maintaining stakeholder relationships during extended setup phases requires transparency and realistic timeline communication, but ensures immediate revenue activation upon facility completion.
Regulatory Excellence as Market Positioning: Achieving international compliance standards (EuGMP) opens premium market segments and justifies value-based pricing in competitive landscapes.
Ready to Build Your Manufacturing or Laboratory Operations?
Whether you are establishing a new manufacturing facility, upgrading existing operations to meet regulatory requirements, or scaling your laboratory capabilities, Terraforme Biosciences brings proven expertise in GMP compliance, quality system development, and operational excellence. Our founder’s direct experience in building, scaling, and successfully exiting a multi-accredited contract testing laboratory provides unique insights into the technical, regulatory, and commercial challenges you face. Book a discovery call to discuss your laboratory development goals and how we can support your path from concept to operational excellence.